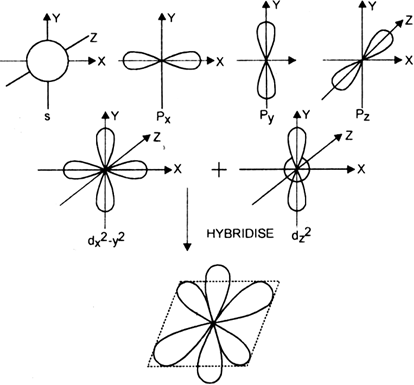
Unless specified, this website is not in any way affiliated with any of the institutions featured. The degenerate hybrid orbitals formed from the standard atomic orbitals: sp hybridization occurs due to the mixing of one s and one p atomic orbital, sp2 hybridization is the mixing of one s and two p atomic orbitals and sp3 hybridization is the mixing of one s and three p atomic orbitals. The five basic shapes of hybridization are linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. The linear combination of a 4 s − orbital with three 4 p − orbitals (4 p x, 4 p y, and 4 p z) and one 3 d − orbital (3 d z 2 ) results in five s p 3 d-orbitals. We have three molecules of iodine here which along with an extra elect… During the process of hybridization, the atomic orbitals of similar energy are mixed together such as the mixing of two ‘s’ orbitals or two ‘p’ orbital’s or mixing of an ‘s’ orbital with a ‘p’ orbital or ‘s’ orbital with a ‘d’ orbital. What is the Hybridization of Sulphur Tetrafluoride? The below diagram will help you depict easily. Sp3: s characteristic 25% and p characteristic 75%. These are directed towards the four corners of a regular, The angle between the sp3 hybrid orbitals is 109.28. The number of hybrid orbitals formed is equal to the number of atomic orbitals mixing. Comments? It forms linear molecules with an angle of 180°. Here 6 will come from sulphur and each of the four fluorine atoms will have 7 electrons. Octahedral: Six electron groups involved resulting in sp3d2 hybridization, the angle between the orbitals is 90°.
#SP3D GEOMETRY FULL#
1 $\begingroup$ What is the difference between $\mathrm$ hybridization? The atomic orbitals of the same energy level can only take part in hybridization and both full filled and half-filled orbitals can also take part in this process, provided they have equal energy.
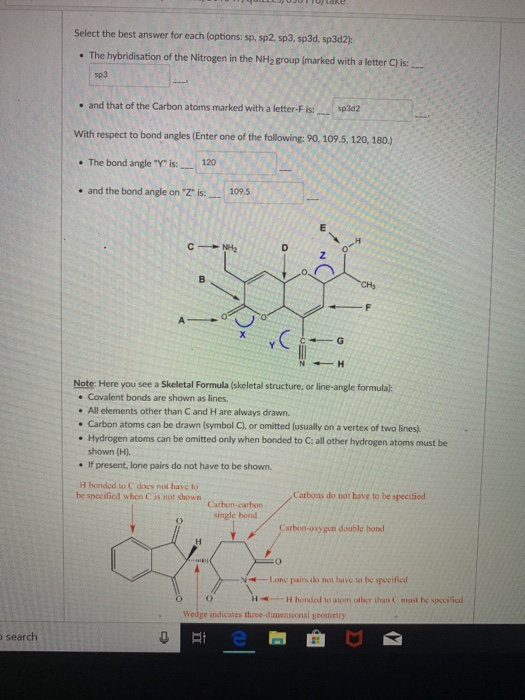
Are they one in the same? School Walla Walla University Course Title CHEM 142 Type.
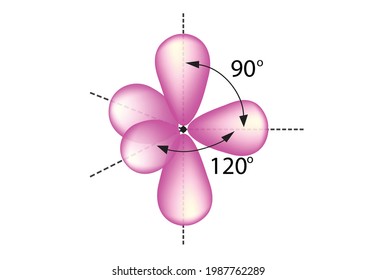
(b) These orbitals combine to form a trigonal bipyramidal structure with each large lobe of the hybrid orbital pointing at a vertex. The repulsion between these groups produce a linear shape for the molecule with bond angle of 180. Sp and sp2 hybridization results in two and one unhybridized p orbitals respectively whereas in sp3 hybridization there are no unhybridized p orbitals. We're sorry, but in order to log in and use all the features of this website, you will need to enable JavaScript in your browser. * Each of these sp3 hybrid orbitals f… Generally, the Lewis structure is helpful to understand the molecular geometry of any given chemical compound. This intermixing is based on quantum mechanics. These hybridizations are only possible for atoms that have d orbitals in their valence subshells (that is, not those in the first or second period). As there are molecules of Iodine, one molecule of Iodinewill be in the centre. These lone pairs will also be accommodated in hybridized orbitals.Sp 3 d Hybridization sp 3 d hybridization is shown in phosphorus penta chloride (PCl 5 ).

Molecular Geometry (three dimensional structure) can be determined by the number of -bonds and the lone pairs on the central atom. pi ()-bonds form by side-ways overlap of unhybridized p- and d-orbitals, weak bonds will from. The basis for the Valence Bond theory: Sigma () bonds form by head on overlap of unhybridized, s-orbital-s-orbital, p-orbital-p-orbital, s-orbital-p-orbital and hybridized, (sp, sp2,sp3, sp3d and sp3d2) orbitals, strong bonds will form. Molecular Geometry, using Orbital Hybridization These notes should not replace the reading of good text books.
